Variation in Body Weights and Immune Response of FUNAAB Alpha and Exotic Broiler Chickens Inoculated with Attenuated Eimeria tenella Vaccine
| Received 12 Jul, 2023 |
Accepted 20 Dec, 2023 |
Published 12 Jan, 2024 |
Background and Objective: Coccidiosis is one of the pathogenic diseases of economic importance to broiler chickens in the tropics. The aim of this study was to investigate the variation in body weight and immune response of FUNAAB Alpha and exotic broiler chickens inoculated with attenuated Eimera tenella vaccine. Materials and Methods: The experiment was laid out in a completely randomized design for a period of 8 weeks. The experimental birds consist of 200 broilers of 2 breeds namely: Three strains of FUNAAB Alpha (normal feather, frizzle feather and naked neck) and one breed of exotic chicken (Arbor Acre). The body weight, haematological parameters and lesion counts were collected and analyzed for the effects of genotype pre and post-inoculation. Results: The Arbor Acre broiler chickens had significantly (p<0.05) heaviest body weight at days 0, 6 and 15. There is variation in haematological responses to E. tenella challenged among the four broiler genotypes with the highest (p<0.05) values of haematological parameters recorded in FUNAAB Alpha broilers. The oocyte counts, caecal and mucosa lesions were higher (p<0.05) in Arbor Acre broilers. Conclusion: Therefore, the FUNAAB Alpha broilers can be selected against coccidiosis diseases for the development of broiler chickens or meat free from protozoan contamination in Nigeria.
| Copyright © 2024 Oguntade et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Coccidiosis has been identified as one of the most economically significant diseases of domestic chicken due to the tremendous morbidity and mortality it causes and also the enormous costs associated with its control1,2. The genus Eimeria which are members of the intracellular parasite are responsible for this intestinal parasitic infection2. There are seven different Eimeria species that can cause coccidiosis in chickens, some of which are more virulent than others. These include E. tenella, E. maxima, E. necatrix and E. brunette, others such as E. acervulina, E. mitis and E. praecox are less virulent2.
The intestinal tracts of various animals and birds are the target of Eimeria parasites and infection from these parasites usually occurs when an animal consumes feed and water that have been contaminated with sporulated oocytes from Eimera organism3. The typical symptoms of coccidiosis, which also has high mortality and morbidity rates, include dysentery, enteritis, diarrhea (which in some cases may be bloody due to the pathogenic Eimeria species), emaciation, a lower feed conversion rate, delayed sexual maturity, drooping wings, poor growth and low production. E. tenella is the most frequent and pathogenic among Eimeria species that impacts the poultry industry globally, with 100% morbidity and a high death rate due to serious injuries to the chickens' digestive4.
The Federal University of Agriculture, Abeokuta, developed the improved indigenous Nigerian chicken known as FUNAAB Alpha. This chicken is an improved breed of Nigerian local chicken breed and was developed through conscientious efforts of scientists at the Department of Animal Breeding and Genetics, Federal University of Agriculture, Abeokuta5. The breed was created using improved naked neck and frizzled feather chicken genotypes over 6 generations of selection and inbreeding, followed by 4 generations of crossbreeding with some exotic lines to enhance its growth and carcass quality5.
In both domestic and foreign breeds of chickens, coccidiosis has been identified as a severe parasitic disease with a significant economic impact4,6. Both exotic (broilers, layers and cockerels) and Nigerian local chickens are susceptible to coccidiosis. However, the host's breed is one of the factors that influence how the host reacts to parasite diseases. Some animal breeds exhibit far greater resistance than others and can endure exposure to infections that would be fatal to other breeds7. This study focused on breed variations in body weight and immune response of improved Nigerian indigenous and exotic broiler chicken inoculated with attenuated Eimera tenella vaccine.
MATERIALS AND METHODS
Experimental site: This research was conducted at the Poultry Unit of the Teaching and Research Farm of Ambrose Alli University, Ekpoma, Edo state. The experiment was laid out in a completely randomized design for a period of 8 weeks between July and September. The experimental birds consist of 200 broiler chickens of 2 breeds namely: Three strains of FUNAAB Alpha (normal feather, frizzle feather and naked neck) and one breed of exotic broiler (Arbor Acre). The FUNAAB Alpha also called improved Nigerian indigenous chicken was sourced from Pearl Farm, Federal University of Agriculture, Abeokuta Ogun State Nigeria while the Arbor Acre was purchased from a reputable hatchery in Ibadan, Oyo State.
Management of experimental birds: The birds were reared from the day old and brooded for a period of 3 weeks during which adequate ventilation, heat and humidity were ensured. The birds were reared in chick cage to prevent them from having contact with their droppings. They were wing-tagged for proper identification and subjected to the same management practices throughout the experimental period of 8 weeks. The birds were allowed access to commercial feeds and clean water ad libitum. The birds were fed starter and finisher mash, each containing 23% and 19% crude protein, from 0 to 4 and 5-8 weeks of age, respectively. To curb the spread of infectious diseases, the broiler chicken vaccination schedule was closely followed and proper sanitation was maintained.
Measurement of body weights: The body weights of the experimental birds were measured at 3 weeks before inoculation (on the inoculation day-day 0) and on days 1, 3, 6, 9, 12 and 15 post inoculation with Eimera tenella vaccine. The body weight was measured in the morning before the birds were fed and it was done with Atom ‘A’ Camry digital scale with 0.01 g sensitivity.
Inoculation with Eimera tenella vaccine: Eimera tenella vaccine was purchased from National Veterinary Research Institute, Vom, in Plateau State, Nigeria. At 3 weeks of age, the experimental birds were inoculated with Eimera tenella vaccine, through oral inoculation at 100,000 (that is 1×105) doses per bird as specified by the manufacturer. All challenged birds were kept in battery cages to prevent them from having direct contact with their faeces.
Blood collection: Blood samples (1 mL) were collected with sterile needle and syringe from the wing veins of the experimental birds two days before inoculated with attenuated Eimera tenella for base line study and also 7 days’ post inoculation. The blood sample collected was transferred into tubes containing Ethylenediaminetetraacetic acid (EDTA) as an anticoagulant and labelled accordingly. The un-coagulated blood was used to determine Packed Cell Volume (PCV) by a microhaematocrit capillary tube using a hematocrit reader as described by Ezebo et al.8. The Red Blood Cell (RBC), White Blood Cell (WBC) and differential counts were analyzed immediately (not exceeding 24 hrs) after collection of blood using an automated cell counter8. Other haematological indices such as Mean Corpuscular Volume (MCV), Mean Corpuscular Hemoglobin (MCH) and Mean Corpuscular Haemoglobin Concentration (MCHC) were calculated8.
Collection of faecal samples: Faecal samples were collected in polythene bags after inoculation from challenged chickens on days 1, 3, 6, 9, 12 and 15. The faecal samples were subjected to a simple floatation method and viewed under an electron microscope (OLYMPUS CH) for parasitological examination. The faecal oocyst load was determined using McMaster egg counting method. Mortality was recorded from day 1 to day 15 post-inoculation. At post-mortem, caecal lesion scores (LES) were assessed according to Johnson and Reid9, from 0 (no lesion) to 4 (most severe lesions). Mortality due to coccidiosis was scored 5. Preliminary analysis was carried out where homogeneity was tested.
Statistical analysis: The data collected on body weight, haematological parameters and lesion counts were analyzed for the effects of broiler genotype pre and post-inoculation. The analysis was done by using Analysis of Variance (ANOVA) using the General Linear Model (GLM) of the SAS 9.4 software (SAS Institute Inc., USA). Differences between means were separated using Duncan’s multiple range and significance was considered at p<0.05.
The model is:
Where, Yij is the parameter of interest, μ is the population mean, Gj is the effect of ith genotype on the parameter of interest (i = 1, 2, 3, 4) and ɛij is the residual error.
RESULTS
Body weights of the experimental broiler as affected by genotype pre and post inoculation with Eimera tenella: The body weights of FUNAAB Alpha and Arbor Acre broiler chickens as affected by genotypes pre (day 0) and post (days 1, 3, 6, 9, 12 and 15) inoculation with Eimera tenella vaccine are shown in Table 1. The Arbor Acre broiler chicken had significantly (p<0.05) highest body weight in all the day’s considered pre and post inoculation with Eimera tenella. However, comparable to the body weights of naked neck broiler chickens at days 1, 3, 9 and 12 post inoculation. The frizzle and normal feather broiler chickens had statistically similar body weights on all the days considered pre and post inoculation.
The haematological profile of improved Nigerian indigenous (normal feather, frizzle feather and naked neck) and exotic (Arbor Acre) broilers before inoculating with Eimera tenella vaccine are shown in Table 2. There were no significant (p>0.05) differences in all the haematological parameters studied except in Red Blood Cells (RBC), Lymphocytes (LYMP) and Mean Corpuscular Volume (MCV). The RBC was significantly (p<0.05) influenced with the highest value (2.71 106 μL–1) in normal feather broiler and the least found in the naked neck including frizzle feather and Arbor Acre broilers.
| Table 1: | Body weights of the experimental broiler as affected by genotype | |||
| Post inoculation days | Genotypes | Body weight |
Sample size |
| 0 | Naked neck | 235.41±8.65b |
50 |
| Frizzle feather | 212.60±8.65c |
50 |
|
| Normal feather | 218.98±8.42c |
50 |
|
| Arbor Acre | 279.91±8.30a |
50 |
|
| 1 | Naked neck | 391.00±8.65a |
50 |
| Frizzle feather | 346.88±8.60b |
50 |
|
| Normal feather | 336.13±8.42b |
50 |
|
| Arbor Acre | 413.80±8.30a |
50 |
|
| 3 | Naked neck | 428.55±19.47a |
50 |
| Frizzle feather | 409.92±19.18b |
50 |
|
| Normal feather | 349.09±18.52b |
50 |
|
| Arbor Acre | 442.49±18.60a |
50 |
|
| 6 | Naked neck | 460.14±28.02b |
50 |
| Frizzle feather | 425.75±24.22b |
50 |
|
| Normal feather | 402.55±21.23b |
50 |
|
| Arbor Acre | 497.59±23.41a |
50 |
|
| 9 | Naked neck | 566.25±20.00a |
50 |
| Frizzle feather | 481.15±20.00b |
50 |
|
| Normal feather | 433.27±19.47b |
50 |
|
| Arbor Acre | 547.69±19.18a |
50 |
|
| 12 | Naked neck | 671.44±46.52a |
50 |
| Frizzle feather | 592.92±53.93b |
50 |
|
| Normal feather | 556.95±52.57b |
50 |
|
| Arbor Acre | 710.74±58.68a |
50 |
|
| 15 | Naked neck | 1057.14±48.52b |
50 |
| Frizzle feather | 1002.75±48.52b |
50 |
|
| Normal feather | 1010.42±47.24b |
50 |
|
| Arbor Acre | 1308.59±46.52a |
50 |
|
| Means in the same column with different superscripts abcSignificantly different (p<0.05) and ±: Plus or minus standard error | |||
| Table 2: | Haematological parameters of improved Nigerian indigenous and exotic (Arbor Acre) broiler chickens before inoculated with Eimera tenella vaccine | |||
| Parameter | Normal feather |
Frizzle feather |
Naked neck |
Arbor Acre |
| PCV (%) | 30.75±6.42 |
31.00±1.79 |
27.50±2.71 |
29.25±3.56NS |
| RBC (106 μL–1) | 2.71±0.67a |
2.55±0.20ab |
2.29±0.14b |
2.46±0.24ab |
| Hb (g dL–1) | 10.30±2.01 |
10.33±0.51 |
9.18±0.92 |
9.98±0.96NS |
| WBC (103 μL–1) | 11.58±3.08 |
9.38±5.69 |
12.03±4.88 |
8.53±1.86NS |
| HET (%) | 28.00±7.39 |
32.00±3.99 |
30.50±2.14 |
30.50±4.28NS |
| LYMP (%) | 68.75±7.50a |
63.75±4.27b |
66.00±3.21ab |
65.50±4.17ab |
| MONO (%) | 1.25±0.65 |
2.00±0.99 |
1.25±0.37 |
1.25±0.99NS |
| EOS (%) | 1.00±0.53 |
1.00±0.65 |
1.00±0.65 |
1.00±0.75NS |
| BAS (%) | 1.00±0.53 |
1.25±0.37 |
1.25±0.83 |
1.50±0.37NS |
| MCV (fL) | 11.45±0.49b |
12.17±0.31a |
11.93±0.48a |
11.82±0.38ab |
| MCH (pg/cell) | 38.21±1.59 |
39.23±0.57 |
38.29±1.05 |
38.84±0.95NS |
| MCHC (g dL–1) | 33.47±0.69 |
33.39±0.21 |
33.34±0.11 |
33.67±0.44NS |
| Mean in the same row with different superscripts abSignificantly different (p<0.05), PCV: Packed Cell Volume, RBC: Red Blood Cell, HB: Haemoglobin, WBC: White Blood Cells, HET: Heterophils, LYMP: Lymphocytes, MONO: Monocytes, EOS: Eosinophils, BAS: Basophils, MCV: Mean Corpuscular Volume, MCH: Mean Corpuscular Hemoglobin, MCHC: Mean Corpuscular Hemoglobin Concentration, NS: Non-significance and ±: Plus or minus standard error | ||||
The LYMP value was highest (p<0.05) in normal feather broilers (68.75%) but similar (p>0.05) to 66.00 and 65.50% obtained in the naked neck and Arbor Acre broilers respectively. The frizzle broiler had the least LYMP (63.75%), although not significantly (p>0.05) different from naked neck and Arbor Acre broilers. The frizzle feather broiler recorded the highest (p<0.05) value of MCV but not significantly (p>0.05) different from the values obtained from naked neck and Arbor Acre broilers, respectively, while the normal feather broiler had the least MCV however similar (p>0.05) to the value obtained from Arbor Acre broiler. Other haematological parameters examined are not significantly (p>0.05) affected by the broiler genotype.
| Table 3: | Haematological parameters of improved Nigerian indigenous and exotic (Arbor Acre) broiler chickens after inoculated with Eimera tenella vaccine | |||
| Parameter | Normal feather |
Frizzle feather |
Naked neck |
Arbor Acre |
| PCV (%) | 39.50±1.14a |
31.80±1.19c |
36.00±1.37ab |
32.75±1.37bc |
| RBC (106 μL–1) | 3.31±0.09a |
3.19±0.24ab |
3.06±0.09ab |
2.80±0.11b |
| Hb (g dL–1) | 13.20±0.34a |
10.83±0.40b |
11.98±0.46b |
11.00±0.45b |
| WBC (103 μL–1) | 11.08±0.75b |
12.85±0.69a |
13.68±0.37a |
10.30±0.55b |
| HET (%) | 35.5O±1.50a |
33.05±1.54ab |
30.25±2.02b |
32.00±0.87ab |
| LYMP (%) | 61.25±1.62 |
64.80±1.29 |
66.00±2.17 |
64.50±0.86NS |
| MONO (%) | 1.00±0.20b |
2.30±0.30a |
1.85±0.20a |
1.25±0.31b |
| EOS (%) | 1.00±0.20ab |
1.55±0.29a |
0.75±0.20b |
1.25±0.20ab |
| BAS (%) | 1.25±0.12 |
0.80±0.35 |
1.25±0.20 |
1.00±0.20NS |
| MCV (fL) | 12.25±0.11 |
12.66±0.38 |
12.42±0.14 |
12.01±0.09NS |
| MCH (pg/cell) | 39.37±0.05 |
39.25±0.37 |
39.15±0.36 |
39.01±0.24NS |
| MCHC (g dL–1) | 33.48±0.14 |
33.36±0.22 |
33.49±0.07 |
33.57±0.16NS |
| Mean in the same row with different superscripts abcSignificantly different (p<0.05), PCV: Packed Cell Volume, RBC: Red Blood Cell, HB: Haemoglobin, WBC: White Blood Cells, HET: Heterophils, LYMP: Lymphocytes, MONO: Monocytes, EOS: Eosinophils, BAS: Basophils, MCV: Mean Corpuscular Volume, MCH: Mean Corpuscular Haemoglobin, MCHC: Mean Corpuscular Haemoglobin Concentration, ±: Plus or minus standard error and NS: Non-significance | ||||
| Table 4: | Effect of genotype on lesion score of Eimeria tenella inoculated broiler chickens genotype | |||
| Genotype | Leison score |
| Normal feather | 3.64±0.33b |
| Frizzle feather | 2.24±0.40b |
| Naked neck | 2.42±0.22b |
| Arbor Acre | 6.45±0.50a |
| Means in the same column with different superscripts abSignificantly different (p<0.05) and ±: Plus or minus standard error | |
Table 3 shows the effect of genotype on the haematological profile of improved Nigerian indigenous (normal feather, frizzle feather and naked neck) and Arbor Acre broilers after inoculation with attenuated Eimera tenella vaccine. The results showed that PCV, RBC, Hb, WBC, HET, MON and EOS were significantly (p<0.05) influenced by the broiler genotype after inoculation with attenuated Eimera tenella vaccine. The normal feather broiler had the highest (p<0.05) PCV (39.50%) followed by similar (p>0.05) value (36.00%) in naked neck broiler, which is also similar (p>0.05) with the value (32.75%) in Arbor Acre while the frizzle feather broiler had the least (31.80%) PCV but was not different (p>0.05) from the value in Arbor Acre broiler. The value of RBC was highest in normal feathers but similar to the values in frizzle and naked neck broilers. The Arbor Acre broiler had the least (p<0.05) value of RBC however not significantly (p>0.05) from the values in frizzle feather and naked neck. The Hb concentration was significantly (p<0.05) affected by the broiler genotype after inoculation with the highest value in normal feather (13.20 g dL–1) followed by 11.98, 11.00 and 10.83 g dL–1 which are similar (p>0.05) in the naked neck, Arbor Acre and frizzle feather broilers respectively. Further, there was a significant (p<0.05) effect of genotype on WBC. The highest (p<0.05) WBC was obtained in the naked neck (13.68 103 μL–1) and frizzle feather (12.85 103 μL–1) broilers while Arbor Acre and normal feather had statistically (p<0.05) similar least values. The HET values were higher (p<0.05) in normal feather broiler (35.50%) but also included (p>0.05) frizzle feather (33.05%) and Arbor Acre broiler (32.00%). The least HET (30.25%) was found in the naked neck, although statistically (p>0.05) the same with the values in the frizzle feather and Arbor Acre broilers. The MONO was affected (p<0.05) by broiler genotype post-inoculation. The MONO followed a similar trend as WBC. The values of EOS obtained post-inoculation was highest (p<0.05) in frizzle feather including (p>0.05) Arbor Acre and frizzle feather broilers while the naked broiler had the least. The LYMP, MCV, MCH and MCHC were not significantly affected by the broiler genotype post inoculation with Eimeria tenella vaccine.
Table 4 shows the effect of genotype on the lesion score of Eimeria tenella inoculated broiler chicken genotypes. The results showed that genotype significantly (p<0.05) influenced the lesion score of improved Nigerian indigenous and exotic broiler chicken with Arbor Acre having the highest (p<0.05) lesion score (6.45) followed by normal feather broiler (3.64) with statistically (p>0.05) similar values of 2.42 and 2.24 in the naked neck and frizzle feather broilers, respectively.
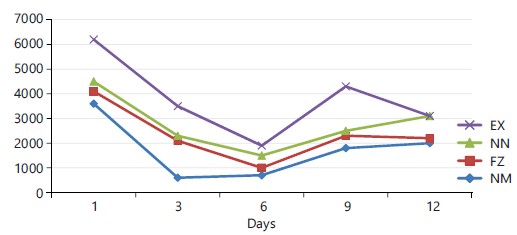
|
Figure 1 shows oocyte counts of improved Nigerian indigenous and Arbor Acre broilers inoculated with Eimera tenella vaccine. The oocyte counts showed three patterns. First, the number of oocytes decreased from day 1 through day 3 to day 6 post inoculations and thereafter increased from day 6 to the peak at day 9 in all the genotypes. The number of oocytes started declining from day 9 through day 12 of post-inoculation.
DISCUSSION
The body weight increases as the birds get older across the genotypes. The highest body weight recorded among the Arbor Acre may be attributed to the fact that they have been long selected for higher body weight and early maturity. However, the naked neck genotype had comparable weights with Arbor Acre broilers on all the day’s considered post-inoculation. This implies a higher concentration of exotic genes in naked neck broilers than in other genotypes of FUNAAB Alpha broilers. This demonstrated that if the naked neck broilers can be further improved and selected, it may successfully substitute Arbor Acre and other exotic broilers in tropical environments such as in Nigeria. Also, the rearing environment and management practices are the same for the four broiler genotypes, therefore differences or similarities in body weight among them could be attributed to genetic differences10. This was similar to the findings of Atansuyi et al.11. In addition, Atansuyi et al.12 reported that Marshall broiler has higher body weight than Nigerian indigenous broiler type chicken.
Hematological parameters have been observed as good indicators of the physiological status of the animal and their changes are important in assessing the response of such animals to various physiological situations13. In the current study, the results of haematological analysis showed that broilers used for the study were not affected by genotypes before inoculation with the Eimera tenella vaccine except in LYMP and MCV. This suggests that the birds were healthy before challenged with Eimera tenella. The present results of PCV and RBC values were in agreement with values given by Adenaike et al.14, where no significant genotype effect was observed in the PCV and RBC of Nigerian local chickens. However, variation in LYMP and MCV among the four genetic groups of broiler in this study may offer explanation for the defence mechanism of the birds. The existence of differences is an indication that the four genotypes can be ranked differently in terms of immunity and productivity. This implies that the birds were in good health prior to being exposed to Eimera tenella. The current PCV and RBC values were consistent with those reported by Adenaike et al.14, who found no significant genetic effect in the PCV and RBC of local Nigerian chickens. However, differences in LYMP and MCV amongst the four broiler genetic groups of broilers in this study may provide a reason for the birds' defense mechanism. These discrepancies suggest that the four genotypes can be ranked differently in terms of resistance to disease and production. All the haematological values obtained before inoculation are within the ranges reported by Islam et al.15 and Simaraks et al.16 in chickens.
The post-inoculation hematological analysis revealed the highest values of PCV, RBC and Hb in a normal feather broiler. This was contrary to the report of El-Safty et al.17 that the naked neck gene improved PCV compared to that of the normal-feathered chickens. However, the higher values of PCV, RBC and Hb in normal feather broilers suggest better physiological functions including feed utilization and cellular respiration. The PCV is the relative quantity of cells compared to the total volume of blood18. The function of RBC is to transport oxygen from the lungs to tissues and remove carbon dioxide from the tissues to the lung in the body via haemoglobin19. Hemoglobin concentration is useful in detecting anaemia in avian species in general20. The variations in these blood parameters among these genetic groups of E. tenella inoculated broilers can be used with other production traits to exploit the selection of improved Nigerian indigenous broilers for coccidiosis disease14. However, the values obtained in all the genotypes fell within the physiological range reported on chickens14-16. Generally, the values of WBC were higher in improved Nigerian indigenous broilers before and post inoculation with the highest values in the naked neck and frizzle feather broilers post-inoculation. The major functions of WBC and its differentials are to fight infections, defend the body by phagocytosis against invasion by foreign organisms and produce or at least transport and distribute antibodies in immune response21. Therefore, the higher and significant increase in WBC in improved Nigerian indigenous broilers before and post-inoculation suggests innate attributes to generate antibodies rapidly and defend the body against pathogenic organisms. Thus, it can be said that the improved Nigerian indigenous broilers are superior to Arbor Acre broilers in terms of survival in the coccidiosis disease environment. This may not be sufficient evidence, though, to categorize the genotypes as resistant or vulnerable to coccidiosis. Additionally, this study's average WBC counts across the various genotypes were consistent with the findings of Emenalom et al.22.
The lower intestine mucosa of the four genotypes contained E. tenella and the genotype had significant effects on the severity of the lesions. However, compared to normal feather, naked neck and frizzle feather broilers, a considerable number of E. tenella oocysts and clusters of big schizonts were seen in the Arbor Acre broiler, even though lesion score was probably not the best measure of immunological response to coccidiosis in this investigation. The caeca of the Arbor Acre broiler had a high quantity of oocysts and significant tissue damage, according to a pathological analysis of the caeca to assess the severity of the E. tenella infection on the broilers in this study. This outcome was consistent with the findings of Sharma et al.23, who noted significant tissue loss, hemorrhage and disruption of the caecal glands. Chanie et al.24 also reported similar in separate studies on E. tenella-infected local and RIR chickens and E. tenella and Eimeria acervulina-infected broiler chickens, respectively.
Despite having a comparative advantage in terms of body weight, this study found that Arbor Acre broilers are relatively susceptible to coccidiosis disease in the tropics. However, variations at the gene level may be necessary to accurately screen both FUNAAB Alpha and Arbor Acre broilers as being tolerant or susceptible to Eimera tenalla organism.
CONCLUSION
The body weights of Arbor Acre and naked neck broilers are comparable but higher than those of normal feather and frizzle feather broilers. There is variation in haematological responses to E. tenella challenged among the four broiler genotypes in this study. The normal feather broiler showed a better response in terms of PCV, RBC and Hb. Generally, all the haematological parameters examined were higher in improved Nigerian indigenous broilers. The oocyte counts and caecal and mucosa lesions were higher in Arbor Acre broilers. The improved Nigerian indigenous broilers especially the naked neck genotype can be selected against coccidiosis in the tropics. However, more factors (such as antibody titre and cytokine levels) can be investigated as coccidiosis immune response traits to ascertain the response of improved Nigerian indigenous and Arbor Acre broilers to coccidiosis.
SIGNIFICANCE STATEMENT
Genetically resistant breed of broiler chicken to coccidiosis have not been reported in tropical environments and yet coccidiosis is an endemic disease of chicken in the tropics. Therefore, the results obtained in this study can form the basis for selection of resistant breed of broiler chicken in the tropics. This study provided insight into the comparable weights of FUNAAB Alpha and Arbor Acre broiler chickens and their immune-competence against coccidiosis. In addition, the information provided on haematology can be used as diagnostic tools for assessing the health of FUNAAB Alpha and Arbor Acre broiler chickens in a coccidiosis prevalent environment.
REFERENCES
- Nematollahi, A., G. Moghaddam and R.F. Pourabad, 2009. Prevalence of Eimeria species among broiler chicks in Tabriz (Northwestern of Iran). Munis Entomol. Zool., 4: 53-58.
- Sharma, S., A. Iqbal, S. Azmi, I. Mushtaq, Z.A. Wani and S. Ahmad, 2013. Prevalence of poultry coccidiosis in Jammu region of Jammu & Kashmir State. J. Parasitic Dis., 39: 85-89.
- Allen, P.C. and R.H. Fetterer, 2002. Recent advances in biology and immunobiology of Eimeria species and in diagnosis and control of infection with these coccidian parasites of poultry. Clin. Microbiol. Rev., 15: 58-65.
- Lawal, J.R., S.M. Jajere, U.I. Ibrahim, Y.A. Geidam, I.A. Gulani, G. Musa and B.U. Ibekwe, 2016. Prevalence of coccidiosis among village and exotic breed of chickens in Maiduguri, Nigeria. Vet. World, 9: 653-659.
- Durosaro, S.O., O.S. Iyasere, D.O. Oguntade, B.M. Ilori and V.J. Oyeniran et al., 2023. Effect of genotype on growth, fear-related behaviours, and walking ability of FUNAAB Alpha broiler chickens. Appl. Anim. Behav. Sci., 267.
- Jatau, I.D., N.H. Sulaiman, I.W. Musa, A.I. Lawal, O.O. Okubanjo, I. Isah and Y. Magaji, 2012. Prevalence of coccidia infection and preponderance Eimeria species in free range indigenous and intensively managed exotic chickens during hot-wet season, in Zaria, Nigeria. Asian J. Poult. Sci., 6: 79-88.
- Owen, J.B. and R.F.E. Axford, 1991. Breeding for Disease Resistance in Farm Animals. C.A.B. International, Wallingford, ISBN: 9780851987101, Pages: 499.
- Ezebo, R.O., J.N. Omumuabuike, B.G. Esimai, E.E. Ilouno, O.U. Obieze, J.N. Obienyem and C.A. Uba, 2021. Effect of protein extract of Mangifera indica pollen on the erythrocytes (red blood cell) of albino rats. World News Nat. Sci., 36: 114-124.
- Johnson, J. and W.M. Reid, 1970. Anticoccidial drugs: Lesion scoring techniques in battery and floor-pen experiments with chickens. Exp. Parasitol., 28: 30-36.
- Ige, A.O., B.R. Rafiu and I.T. Mudasiru, 2016. Effect of genotype on growth traits characteristics of two commercial broiler chickens in a derived Savannah Zone of Nigeria. Int. J. Res. Stud. Agric. Sci., 2: 26-32.
- Atansuyi, A.J., I.O. Olaseinde and C.A. Chineke, 2018. Estimation of body weight from linear body measurements in four-chicken genotypes using linear and quadratic functions. J. Sustainable Technol., 9: 14-24.
- Atansuyi, A.J., C.O. Lasore and C.A. Chineke, 2017. Growth performance characteristics and linear body measurements of four-chicken genotypes raised under intensive management system in South-Western Nigeria. Appl. Trop. Agric., 22: 122-127.
- Oguntade, O.D., B.M. Ilori, O.S. Durosaro, A.J. Abiona, C.E. Isidahomen and M.O. Ozoje, 2021. Genetic variations in haematological indices of local and exotic Turkeys inoculated with attenuated Salmonella typhimurium vaccine. Asian J. Biochem., Genet. Mol. Biol., 8: 19-29.
- Adenaike, A.S., A.O. Mabunmi, M.I. Takeet, O.D. Adenaike and C.O.N. Ikeobi, 2016. Genetic differences in the body weight and haematological traits of Nigerian indigenous chickens infected with Eimeria tenella. Trop. Anim. Health Prod., 48: 1443-1447.
- Islam, M.S., N.S. Lucky, M.R. Islam, A. Ahad and B.R. Das et al., 2004. Haematological parameters of Fayoumi, Assil and local chickens reared in Sylhet Region in Bangladesh. Int. J. Poult. Sci., 3: 144-147.
- Simaraks, S., O. Chinrasri and W. Aengwanich, 2004. Hematological, electrolyte and serum biochemical values of the Thai indigenous chickens (Gallus domesticus) in Northeastern, Thailand. Songklanakarin J. Sci. Technol., 26: 425-430.
- El-Safty, S.A., U.M. Ali and M.M. Fathi, 2006. Immunological parameters and laying performance of naked neck and normally feathered genotypes of chicken under winter conditions of Egypt. Int. J. Poult. Sci., 5: 780-785.
- Nwosu, C.O. and M. Stephen, 2005. Parasites and associated changes in packed cell volume of horses (Equus caballus) in the semi-arid zone, North Eastern Nigeria. Anim. Res. Int., 2: 329-331.
- Odunitan-Wayas, F., U. Kolanisi and M. Chimonyo, 2018. Haematological and serum biochemical responses of ovambo chickens fed provitamin A biofortified maize. Braz. J. Poult. Sci., 20: 425-434.
- Minias, P., 2015. The use of haemoglobin concentrations to assess physiological condition in birds: A review. Conserv. Physiol., 3.
- Z.M. Osman, A.M. Gomaa, H.M. Hussein, F.H.S. Soliman and A.F. El-Shobaki, 2004. Association between retinol metabolism and diabetic retinopathy. Pol. J. Food Nutr. Sci., 54: 391-396.
- Emenalom, O.O., B.O. Esonu, E.B. Etuk and C. Anaba, 2009. Effect of Mucuna pruriens (velvet bean) leaf meal on performance and blood composition of finisher broiler chickens. Niger. J. Anim. Prod., 36: 52-60.
- Sharma, S., S. Azmi, A. Iqbal, N. Nasirudullah and I. Mushtaq, 2015. Pathomorphological alterations associated with chicken coccidiosis in Jammu Division of India. J. Parasitic Dis., 39: 147-151.
- Chanie, M., T. Negash and B.S. Tilahun, 2009. Occurrence of concurrent infectious diseases in broiler chickens is a threat to commercial poultry farms in Central Ethiopia. Trop. Anim. Health Prod., 41: 1309-1317.
How to Cite this paper?
APA-7 Style
Oguntade,
D.O., Isidahomen,
C.E., Utulu,
G.G., Ekinadose,
O., Ilori,
B.M., Durosaro,
S.O., Loyinmi,
I. (2024). Variation in Body Weights and Immune Response of FUNAAB Alpha and Exotic Broiler Chickens Inoculated with Attenuated Eimeria tenella Vaccine. Research Journal of Veterinary Sciences, 17(1), 8-16. https://doi.org/10.3923/rjvs.2024.08.16
ACS Style
Oguntade,
D.O.; Isidahomen,
C.E.; Utulu,
G.G.; Ekinadose,
O.; Ilori,
B.M.; Durosaro,
S.O.; Loyinmi,
I. Variation in Body Weights and Immune Response of FUNAAB Alpha and Exotic Broiler Chickens Inoculated with Attenuated Eimeria tenella Vaccine. Res. J. Vet. Sci 2024, 17, 8-16. https://doi.org/10.3923/rjvs.2024.08.16
AMA Style
Oguntade
DO, Isidahomen
CE, Utulu
GG, Ekinadose
O, Ilori
BM, Durosaro
SO, Loyinmi
I. Variation in Body Weights and Immune Response of FUNAAB Alpha and Exotic Broiler Chickens Inoculated with Attenuated Eimeria tenella Vaccine. Research Journal of Veterinary Sciences. 2024; 17(1): 8-16. https://doi.org/10.3923/rjvs.2024.08.16
Chicago/Turabian Style
Oguntade, David, Oluwafemi, Clement Ebanehitah Isidahomen, Gbayi Godwin Utulu, Orose Ekinadose, Babatunde Moses Ilori, Samuel Olutunde Durosaro, and Imisi Loyinmi.
2024. "Variation in Body Weights and Immune Response of FUNAAB Alpha and Exotic Broiler Chickens Inoculated with Attenuated Eimeria tenella Vaccine" Research Journal of Veterinary Sciences 17, no. 1: 8-16. https://doi.org/10.3923/rjvs.2024.08.16

This work is licensed under a Creative Commons Attribution 4.0 International License.



